|
The atomic number of hydrogen atom is 1 so it has only one shell and every orbital in this shell have the same energy. 
This single electron will be present in specific energy level. This transition is an emission process.Ī hydrogen atom has one proton and one electron. When an electron undergoes a transition from higher energy level to lower energy level then it emits energy to complete the process. This transition is an absorption process. When an electron undergoes a transition from lower energy level to the higher energy level then it requires energy to complete the process. Since we didn't have to modify either of the the first 2 reactions, you can add their original enthalpies as given:ĭark lines are observed in the absorption spectrum of hydrogen atom.Īn electronic transition is a process that occurs when an electron undergoes emission or absorption from one energy level to another energy level. To find the enthalpy of the unknown reaction, add the enthalpies of the reactions that work. In fact, it makes things more difficult because it contains N2O, which cannot be cancelled in any way with the reactions we're given. This gives us the unknown reaction so we actually don't need to use reaction (3) at all to answer this question. The 2 NO cancels out and the individual O2's add together to get 2O2. If you add these two reactions together, you'll get: 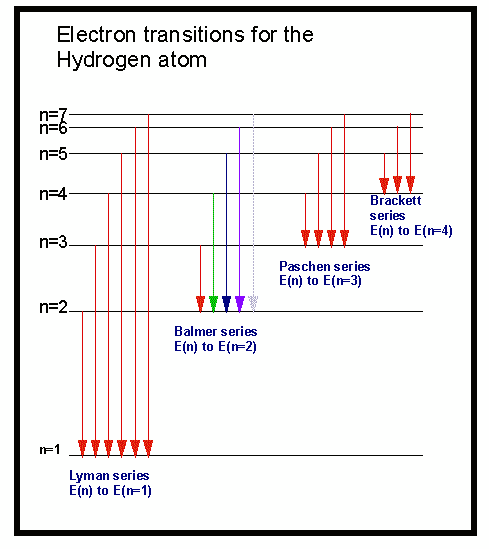 
This means reaction (1) can stay as it is as well. There's 2NO in reaction (1), and it's on the product side, so it will cancel the 2NO on the reactants side of reaction (2). However, it has 2NO on the reactant side - that's not in our unknown reaction, so we need to get rid of it somehow. Looking at what we're given, reaction (2) already has 2NO2 on the product side just like our unknown, so let's start there - it can stay as is. You want to rearrange the given reactions to match the reaction with the unknown enthalpy.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
